Phan Thi Hien, "Esophagus-stomach-duodenum endoscopy in children", Medical Publishing House, Hanoi, Jan 2019 (page 71-90). Extract from specialty book: “NỘI SOI THỰC QUẢN – DẠ DÀY – TÁ TRÀNG TRẺ EM”, Nhà xuất bản y học, Hà Nội, ISBN: 978-604-66-3546-8.
INTRODUCTION
The images of abnormal esophagus are detected by endoscopy mostly related to specific pathology. In which, a common disease is gastroesophageal reflux (GER), especially in children with breast feeding. This disease can lead to ulcerative esophagus, even esophageal stricture and cancer, although they are in children. Beside that, eosinophilic esophagitis due to the increase of eosinophils has commenced receiving attention in recent years. Other pathologies are rare such as infectious esophagitis... Foreign body and esophageal stenosis are presented carefully in the interventional endoscopy part. Based on endoscopic technique, esophageal lesions are clearly observed, simultaneously with mucosal biopsy during endoscopic process has contributed high value in diagnosis as well as in following treatment of these pathologies.
1. ESOPHAGITIS
1.1. Esophagitis associated with gastroesophageal reflux disease
Esophagitis due to refluxis the most common esophageal lesion on endoscopy in children. In medical literature, there are some classifications of reflux esophagitis but they are not significantly different. According to a recommendation of the North America Association for Pediatric Gastroenterology, Hepatology and Nutrition in 2009, visible breaks in the esophageal mucosa are the endoscopic sign of greatest [1]. In clinical practice, many cases with limited lesion such as edema, erythema, pseudopolyp inflammation and pseudomembrane without mucosal breaks. Therefore, at the Endoscopy Department in The Vietnam National Children’s Hospital, we prefer to apply Hetzel-Dentro Modified Los Angeles classification for reflux esophagitis because they are appropriate in describing detail lesions and in application into treatment. The report of Phan Thi Hien et al, 2014 also had used Hetzel-Dent classification, the result had shown 64,7% of esophagitis in reflux children [2].
.jpg)
Hetzel-Dentclassification of esophagitis [3]
.jpg)
Esophagitis grade 2 according to Hezel-Dent classification
.jpg)
Esophagitis grade 4 according to Hezel-Dent classification
In adults, esophagitis is mainly determined by Los Angeles classification with more severe lesions [1].
Esophagitis according to Modified Los Angeles classification:
- Level N: Normal mucosa.
- Level M: Minimized lesion at Z line without mucosal break.
- Level A: One or more mucosal breaks, each ≤ 5 mm in length.
- Level B: At least one mucosal break > 5 mm long, but not continuous between the tops of adjacent mucosal folds.
- Level C: At least one mucosal break that is continuous between the tops of adjacent mucosal folds, but which is not circumferential (< 75% of luminal circumference).
- Level D: Mucosal break that involves at least 75% of the luminal circumference [4].
.jpg)
Mallory Weiss on endoscopy [5]
In severe cases of GER can appear Mallory-Weiss images which occur due to secondary reflux, rarely appearing in children. This is esophageal bleeding from a mucosal laceration, the consequence of exceeding vomiting. This lesion also easily appears if there is portal hypertension in children.
1.2. Eosinophil esophagitis
Eosinophilic esophagitiscan appear at any ages, often associated with allergic
history of patients or their family. Eosinophilic esophagitis is characterized by prominent eosinophil infiltrate of esophageal mucosa, which results in symptoms very similar to that of gastroesophageal reflux disease, more notably vomiting and failure-to-thrive in infants, dysphagia and food impaction in older children and adults [6]. Treatment is based, either on dietary measures such as an elemental formula in small infants and an exclusion diet in children, or pharmacological intervention such as topical or (lees common) systemic steroid.
.jpg)
The lineal furrows sign
Each therapy can achieve a good clinical and histological response but symptoms – and the related eosinophilic infiltrate of esophageal mucosa – tend to recur following treatment withdrawal, so long-term treatment is a major problem [6].
.jpg)
The lineal furrows sign and raised white specks
.jpg)
Circular folds “Trachealization”
Eosinophilic esophagitiscan be characterized by a number of macroscopic abnormalities of the esophagus, including longitudinal furrowing and/or shearing of the mucosa “Crêpe paper” appearance of the mucosa, raised white specks and/or whitish exudates on the mucosal surface, or transient of fixed rings (so – called “trachealization”) of the esophagus, Schatzki’s ring, stricture, felinization, friability and edema of the mucosa [6].
Biopsy principles in eosinophilic esophagitis:
- Mucosal specimens should be fixed in the preservatives other than Bouin’s as this may result in reduced identification of eosinophils [6].
- Biopsies should be taken from both the distal and proximal esophagus, and especially from areas of macroscopic abnormalities, at least 4 pieces.
- Finally, it is also always advisable to take biopsies from stomach andduodenumirrespective of their appearance, in order to rule out – in the appropriate clinical context – eosinophilic gastroenteritis or other causes of gastrointestinal eosinophilia such as drugs, parasites and Crohn’s disease [6].
According to a recent consensus produced by an expert panel of pediatric and adult gastroenterologist, a number of > 15 eosinophils per high power field (40x) in at least one biopsy is required to confirm the diagnosis of eosinophilic esophagitis if without response for high dose of protonpomp inibitorand or normal pH study. Eosinophils are more often spread within the esophageal epithelium,but sometimes they are clustered on theepithelialsurface, resulting of eosinophilic abscess, and this is more often found where raised white specks or whitish exudates are seen at endoscopy[6].
1.3. Infectious esophagitis
Infectious esophagitis is a frequent cause of dysphagia and odynophagia in immunocompromised children. The common types of infectious esophagitisare candida, cytomegalovirus and herpes simplex virus. Endoscopy with brush cytology, biopsy and tissue culture is the most reliable diagnostic method [6].
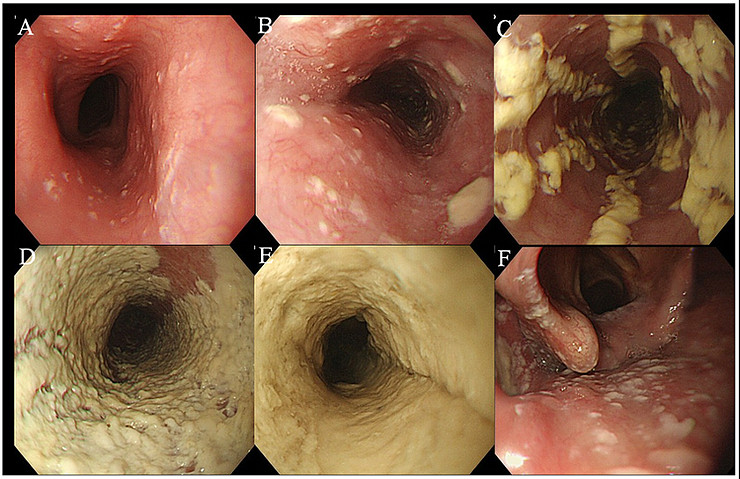
Endoscopic severity of Kodsi's grading [7]
- A: Grade I, a few raised white plaques up to 2 mm in size without edema or ulceration.
- B: Grade II, multiple raised white plaques greater than 2 mm in size without ulceration.
- C: Grade III, confluent, linear, and nodular elevated plaques.
- D: Grade IV, finding of grade III with increased friability of the mucous membranes and occasional narrowing of the lumen.
- E: “White carpet” appearance, thick white plaque cover on esophageal mucosa circumferential narrowing the lumen.
- F: Oral Candidiasis, in which endoscopy can detect laryngopharyngeal candidiasis [7].
Candida esophagitis may present erythematous mucosa covered by scattered or confluent white, cream-colored, thick plaques with high concentration in the distal esophagus [6], these plaques strictly adhere on the mucosal surface. The diagnosis of candida esophagitis is based on Pap tests or culture of samples from brushing techniques. The endoscopic image of esophagitis associated with Cytomegalovirus (CMV) is characterized by well circumscribed ulcerations rounded by normal mucosa. Deep ulcers can see at the middle or distal esophagus. Histological sign is basophilic, intracellular inclusions, a clear halo surrounding the nucleus and presence of multiple smaller periodic is intracytoplasmic inclusions positive with acid Schiff [6].
Herpes simplex virus can begin with herpetic lesions on the lips or buccal mucosa, then odynophagia or dysphagia. Endoscopic diagnosis is based on ulcers (similar to aphthous ulcer in mouth) with raised erythematous margin and grey or yellow base. These ulcers commonly appear at the middle or upper esophagus. In advance disease, may have large, confluent ulcers with yellow exudates at the base. Biopsy from ulcer margin is necessary to take as a viral cytopathic effect. Histological analysis detects typical Cowdry intranuclear inclusions bodies, multinucleate giant cells, ballooning of cells, and chromatin [6].
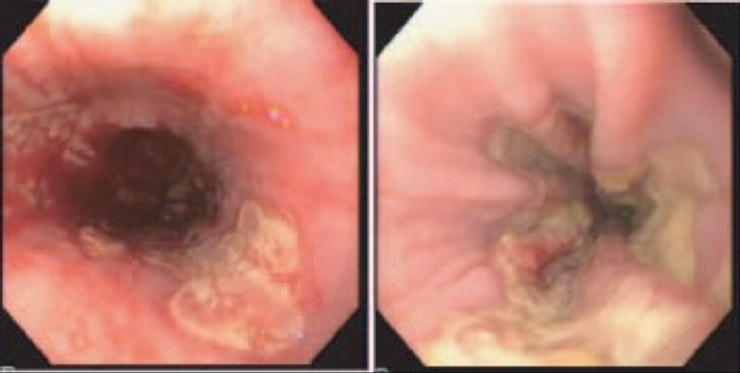
Herpetic esophagitis [8]
1.4. Caustic ingestion
Although the warning is available, but caustic ingestion still happen and frequently occur as tragic accidents in children under 5 years of age or suicide in children over 10 years of age. Alkalis ingestion makes severe injury primarily in the esophagus, strong acid can cause diffuse lesions in the stomach and in esophagus. Sodium hydroxide leads to quickly liquefaction necrosis with deep lesions (all length of the esophagus). Respiratory failure is caused by esophageal perforation or periesophageal inflammation, then mediastinal inflammation or peritonitis are the most serious complications in a short time after swallowing caustic substances [6].
Immediate and prolong-term outcomes are directly related to burn level. Esophageal and gastric lesions can occur in absence of lesions on lips, oral or pharyngeal mucosa. After the patient is in a stable, upper gastrointestinal endoscopy with anesthesia should be performed to evaluate lesions. Mild lesions on the surface by chemical ingestion include erythema, edema with minimal mucosal peeling. The more severe lesions manifest sloughing, hemorrhagic mucosa, mucosal island o ulcer. The severe esophageal lesions are the presence of necrosis or depth ulcer. Almost cases, esophageal mucosa is still observed in the lumen, esophageal spasm with transmural edema leads to pinch and esophageal stenosis in severe cases. Endoscopy should not be indicated or delayed in severe esophagitis by caustic ingestion [6].
Children with an acid-based caustic ingestion are common to more severe lesions at stomach and duodenum, increasing the risk of perforation and fibrosing gastropathy. Children without lesions or with grade 1 lesions can be discharged home, with grade 2 or higher they must be hospitalized for at least 24-48 hours [6].
The patient with moderate or severe burns needed gastrointestinal fasting, parenteral nutritional, broad-spectrum antibiotic and corticoid. Esophageal dilatation is indicated when stenosis appears [6], oftenly after 1 month. If persistent esophageal stenosis, medical treatment is not efficacy, surgical consultation and gastrostomy for feeding until esophageal reconstruction can be done.
Flexible endoscopy can be used to observe stomach event severe caustic ingestion but must be carefully. A study presents that if children without symptoms in alkalis ingestion incident, so, there are no risks of complication and endoscopic indication is not necessary. However, almost all authors believe that early endoscopy is still needed for prognosis. Esophageal lesion grades are evaluated below:
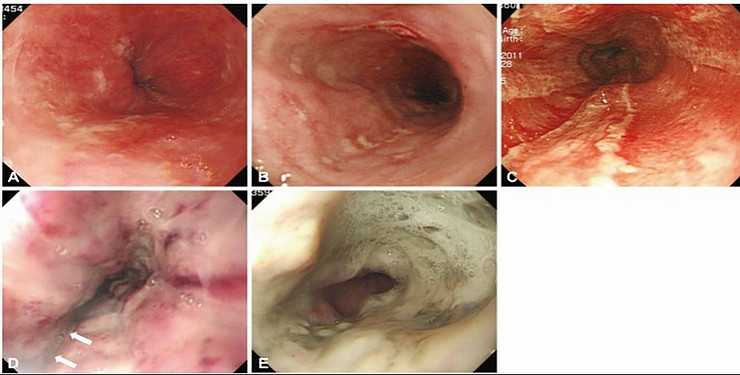
Endoscopic grading of the caustic gastrointestinal injury.
- (A) Grade 1 indicates only slight swelling and redness of the mucosa.
- (B) Grade 2A indicates the presence of superficial ulcers, bleeding, and exudates.
- (C) Grade 2B indicates local or encircling deep ulceration.
- (D) Grade 3A indicates focal necrosis. White arrows indicate focal necrosis.
- (E) Grade 3B indicates extensive necrosis [9].
Grade 3 or grade 2 lesions with whole circumference can lead to esophageal stenosis after 3-6 weeks, it is necessary for esophageal dilation indication [10].
2.ESOPHAGEAL METAPLASIA
2.1. Barrett’s esophagus
Barrett’s esophagus is a chronic complication due to reflux with the malignant risk. However, this lesion is rare in infants. American College of Gastrointestinal Association defines Barrett esophagus as “a change in the esophageal epithelium of any length that can be recognized at endoscopy and confirmed to have intestinal metaplasia by of the tubular esophagus and excludes of intestinal metaplasia of the cardia”. Intestinal metaplasia is easily detected by enough biopsy pisces and suitable time for samples [6].
There are 3 epithelial groups that can be found at esophagus in a patient with chronic gastrointestinal reflux. However, only intestinal metaplasia has dysplasia risk. Intestinal metaplasia is characterized by a change in mucosa about morphologically, and histochemically like small or large intestine mucosa [6].
Other diseases can combine with Barrett’s esophagus such as cerebral palsy, chronic pulmonary disease (especially cystic fibrosis) and after surgery of the esophagus astreia. In adults obesity and obesity at young age have a high risk of Barrett and esophageal adenocarcinoma. We need to mark that Barrett lesion spread in the esophageal circumferential direction easily made a misunderstanding with sliding hernia.
When endoscopy suggests Barrett’s esophagus, need biopsy at least 4 random spicements at each 1-2cm of esophageal mucosa and additional specimens from any mucosal lesion. However, recent researches suggest acid mucus can normally appear at glandular epithelium located in cardiac and distal esophagus without intestinal metaplasia [6].
2.2. Gastric inlet patch in esophagus
is a salmon colored patch that is usually
located just distal to the upper esophageal sphincter.
is a salmon colored patch that is usually
located just distal to the upper esophageal sphincter.
Endoscopic characteristics include a salmon colored patch located in one third of upper esophagus or just below the upper esophageal sphincter, can appear at any age with a prevalence from less 1-18%. The nature is the glandular epithelium of the cardia or body. Almost all children have no clinical symptoms, however some cases can have ulcer, perforation, metaplasia, dysphagia and even cancer. Beside that, children can have extraintestinal symptoms such as cough, wheezing or asthma. Argon plasma is an effective treatment method [11].
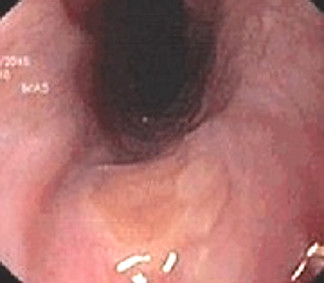
Gastric inlet patch in proximal esophagus
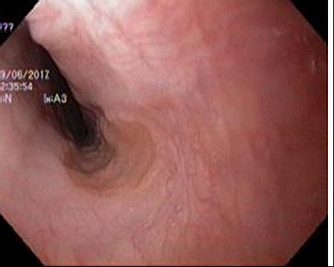
Gastric inlet patch in proximal esophagus
3. ANATOMICAL ABNORMALITIES
3.1. Esophageal stricture and Achalasia
Reflux esophageal stenosis is usually short and located at distal esophagus. These cases appear as white, crescent-like scar or edematous mucosa [6], esophageal stricture often appears in patients with severe reflux, especially in children with cerebral palsy [10].
Schatzki ring is considered short esophageal stenosis, located just above the Z line and surrounded by normal esophageal mucosa.
Barium esophagogram in children with difficult swallowing of solid foods can detect Schatzki although negative endoscopy [6].
Severe stenosis at the middle esophagus is rare, and can be a tracheobronchial sign. This stricture group oftenly is opposite or longer compared to stricture due to esophagitis. Suggested signal of diagnosis is white cartilage without inflammation images on endoscopy [6].
Anastomotic stricture after esophageal atresia surgery is a common complication and indication for esophageal dilatation in children. Main clinical symptoms are vomiting and difficulty swallowing. X-ray shows the image of esophageal stenosis at anastomosis and bigger esophagus above stenosis.
Achalasia is primary esophageal motility disorder, features with lacking distal esophageal peristalsis and incomplete lower esophageal sphincter relaxation. Prominent clinical symptoms are vomiting, dysphagia, lose weight and sometimes chest pain, odynophagia, respiratory signs or regurgitation at night. Unprepared X-ray presents the air/fluid levels in the esophagus and a little gas in the stomach. Barium esophagogram presents the distal esophagus is narrow, the esophagus above narrowing is enlarged, lacking peristalsis. Endoscopy shows closed cardia.
3.2. Sliding hiatal hernia
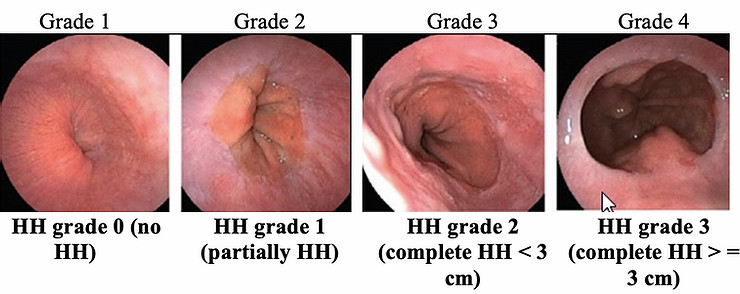
Grading classification of hiatus hernia according to the modified Makuuchi classification (HH grade) measured by the scale on the endoscopic shaft [4]
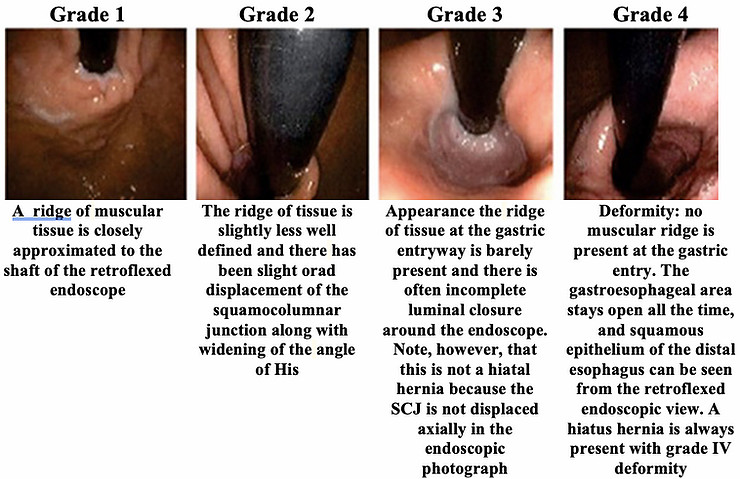
The retroflexed endoscope in hiatus hernia [12]
Endoscopic finding of hiatus hernia is cephalad displacement of Z line by 2 cm or more above the diaphragmatic notch. Diagnosis requires exactly because diaphragm movement depends on respiratory movements: closure during inspiration and opening with expiration. These movements are not clear in the sedated patient, it is difficult to detect during endoscopy. Observation of the cardia with retroflexion technique helps to locate the diaphragmatic notch and clarify the relationship with the Z line [6]. Needed to perform an esophagram to confirm the diagnosis.
3.3. Zenker diverticulum
Zenker diverticulum is common in older patients, it reduces the ability to swallow food (both liquid and solid foods), even in benign cases. Big diverticulum can lead to cricopharyngeal muscle spasms [13]. This lesion is due to the mucosal layer and submucosal layer coming out at just above cricopharyngeal muscle (Killian triangle). Main clinical symptoms are dysphagia and vomiting. Treatment indication when patients have clinical symptoms by surgery [14]. Recently, a flexible endoscopy can treat Zenker diverticulum; it has shown more efficacy and safety than surgery [13,14].
The diverticulum is located at the cervical esophagus where it contains food and leads to infection and perforation. Hence, the esophagus must be clearly observed by endoscopic procedure to avoid perforation.
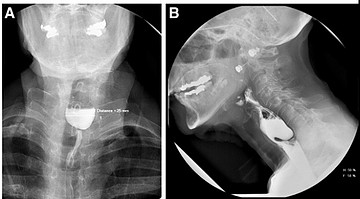
Radiographic images of ZD. (A) Frontal view; (B) lateral view in Zenker diverticulum [15]
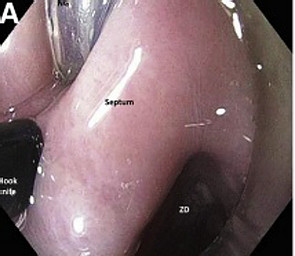
Flexible endoscopic cricopharyngeal myotomy for ZD therapy. Endoscopic view with short, clear cap attached to endoscope tip. Nasogastric tube is in place, and hook knife sheath is seen [15]
4. ESOPHAGEAL VARICES
Bleeding due to esophageal varices is common cause in severe gastrointestinal bleeding in children and accounts for the third of total number of death patients related to cirrhosis in adults. In contrast, extrahepatic portal vein obstruction causes esophageal varices more common in children. Mortality of these patients is related to break of esophageal varices, liver function failure and infection [10].
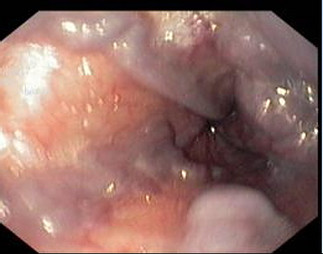
Classification of esophageal varices in 4 grades [10]:
- Grade I: varises disappear with air insufflation.
- Grade II: nonconfluent varices remain identical with air insufflation.
- Grade III: confluent varices cause an obstruction of esophageal lumen.
- Grade IV: addition in grade III: red signs (cherry red sports, red wale markings, hematocystic sports, telangiectasia, and varices overling varices).
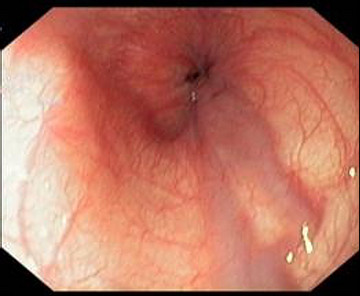
Esophageal varices in grade I
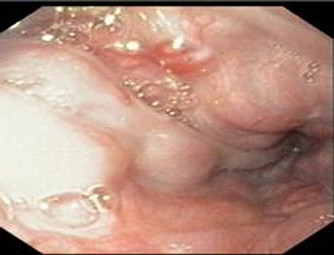
5. FOREIGN BODY IN ESOPHAGUS
Coins are the common foreign body (account for 80%) in children. Almost foreign bodies pass through the entire gastrointestinal tract without any problems. In some cases, obstruction or perforation can appear, these foreign bodys are oftenly at cervical esophagus [10].
Severe clinical conditions are common acute. Flexible endoscopy is an optimized method of intervention.
REFERENCES
1. Vandenplas Y, Rudolph CD, Lorenzo CD et al (2009), “Pediatric Gastroesophageal Reflux Clinical Practice Guidelines”, Journal of Pediatric Gastroenterology and Nutrition, 49(4), 498-547.
2. Phan Thị Hiền, Nguyễn Gia Khánh, Nguyễn Thanh Liêm (2014), “Đánh giá kết quả điều trị bệnh trào ngược dạ dày thực quản ở trẻ em”, Tạp chí khoa học tiêu hóa Việt Nam, 35 (9), 2243-2249.
3. Goldani H, Nunes D, and Ferreira CT, (2012), “Managing gastroesophageal reflux disease in children: The role of endoscopy”, World J Gastrointest Endosc, 4(8), 339-346.
4. Mori A, Ohashi N, Yoshida A et al (2011,) “Unsedated transnasal ultrathin esophagogastroduodenoscopy may provide betterdiagnostic performance in gastroesophageal reflux disease”, Diseases of the Esophagus, 24, 92–98.
5. Na S, Ahn JY, Jung KW et al (2017), “Risk Factors for an Iatrogenic Mallory-Weiss Tear Requiring Bleeding Control during a Screening Upper Endoscopy”, Gastroenterology Research and Practice, 1-6.
6. Gershman G (2012), “Diagnostic upper gastrointestinal endoscopy”, Practical pediatric gastrointestinal endoscopy, (2), 41-81.
7. Nishimura S, Nagata N, Shimbo T et et al (2013), “Factors Associated with Esophageal Candidiasis and Its Endoscopic Severity in the Era of Antiretroviral Therapy”, PLoS One, 8(3), e58217.
8. Gershman G and Marvin Ament (2007), “Diagnostic Upper Endoscopy Technique”, Practical pediatric gastrointestinal endoscopy, (1), 60-101.
9. Park KS (2014), “Evaluation and Management of Caustic Injuries from Ingestion of Acid or Alkaline Substances”, Clinical Endoscopy; 47(4): 301-307.
10. Victor LF (2008), “Gastrointestinal Endoscopy”, Pediatric gastrointestinal desase, 2(1), 1259-1348.
11. Chong VH (2013), “Clinical significance of heterotopic gastric mucosal patch of the proximal esophagus”, World J Gastroenterol, 19(3), 331–338.
12. Kahrilas PJ, Kim HC, and Pandolfino JE (2008), “Approaches to the Diagnosis and Grading of Hiatal Hernia”, Best Pract Res Clin Gastroenterol, 22(4), 601–616.
13. Repici A (2010), “Endoscopic Treatment of Zenker Diverticulum”, Gastroenterol Hepatol, 6(10), 628–630.
14. Bizzotto A, Iacopini F, Landi R et al (2013), “Zenker's diverticulum: exploring treatment options”, Acta Otorhinolaryngol Ital, 33(4), 219–229.
15. Law R, Katzka DA and Baron TH (2014), “Prespectives in clinical gastroenterology and hepatology: Zenker diverticulum”, clinical Gastroenterology and Hepatology, 12(11), 1773-1782.
-

Self-design suction tool
20-05-2021 -

Removing phytobenzoar in Pig's stomach
20-05-2021 -

Remove twisting of the pig colon
04-05-2021 -
Pig stomach endoscopy
04-05-2021
-

Management of Ingested Foreign Bodies in Children: A Clinical Report of the NASPGHAN Endoscopy Committee
28-04-2021 -

Management of Familial Adenomatous Polyposis in Children and Adolescents: Position Paper From the ESPGHAN Polyposis Working Group
28-04-2021 -

Pediatric Colonoscopic Polypectomy Technique
28-04-2021 -

Gastrostomy Placement in Children: Percutaneous Endoscopic Gastrostomy or Laparoscopic Gastrostomy?
28-04-2021







